Model of the tracheo-bronchial region
Computational modeling of the tracheo-bronchial region can provide in-depth knowledge into how the human lung functions mechanically under artificial ventilation. In our studies, we utilize CT-based patient-specific geometries up to a maximum of seven generations. These geometries are extracted from standard CT-images (with a resolution of 0.6mm) which are routinely performed in the hospital. Following segmentation, high quality meshes are generated, typically containing approximately 2 million tetrahedral elements equating to about 1.6 million degrees of freedom. Some further aspects of our computational model are summarized briefly in the following.
Fluid-structure interaction
Considering the effect of fluid-structure interaction (FSI) is important for a better reproduction of the in vivo fluid mechanics in the tracheo-bronchial region. Besides, it is also indispensable for the determination of stresses and strains in the airway wall, which potentially are associated with the onset of inflammation and remodelling of the epithelium. In our model, we take into account both the flexibility of the airway walls and the effect of the surrounding lung parenchyma. To enable a robust and efficient coupling of incompressible flows and soft tissue, we have developed new coupling schemes for FSI which are particularly suited for such complex biological problems.
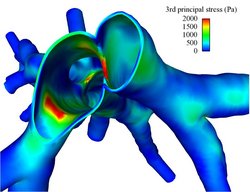
Impedance boundary conditions
Due to limitations on the number of vessels visible on the CT scan, only a part of the airway tree can be resolved in 3D. Therefore, realistic boundary conditions need to be applied at the outlets of the 3D domain in order to take the peripheral region into account. For this purpose, we have developed a reduced-dimensional model of the non-imageable vessels. Briefly, the 3D airway model is supplemented by simplified 1D trees attached to every 3D outlet. By considering the unresolved peripheral impedances, reasonable outflow boundary conditions are derived for the resolved 3D domain.
Turbulence
Despite the plethora of literature related to lung mechanics, there is still debate about the effect of turbulence in the upper airways in the lung community, in particular, how turbulence is generated through the complex laryngeal region affects flow and pressure in the bronchial airways. To investigate pulsatile turbulent flow dynamics of the upper and lower pulmonary airways, an advanced computational approach particularly developed for turbulent flow simulations in complex geometries, the algebraic variational multiscale-multigrid method (AVM3), is utilized. Our investigation suggests that turbulent effects in the bronchial airways are rather weak and can completely decay as early as the third generation, depending on geometry and flow distribution.
Nano-particle mass transfer
Although developed against the background of ventilator-associated lung injuries (VALI), our models are also suitable for numerous other challenging applications associated with the respiratory system. Particularly, nanoparticle deposition in the human lung is of considerable interest, for instance, in case of targeted drug delivery. To investigate the distribution of nanoparticles in the lung, the time-dependent incompressible Navier-Stokes equations are coupled with a transport equation describing the nanoparticle mass transfer.
Publications
Please find publications on our computational model of the tracheo-bronchial region here.